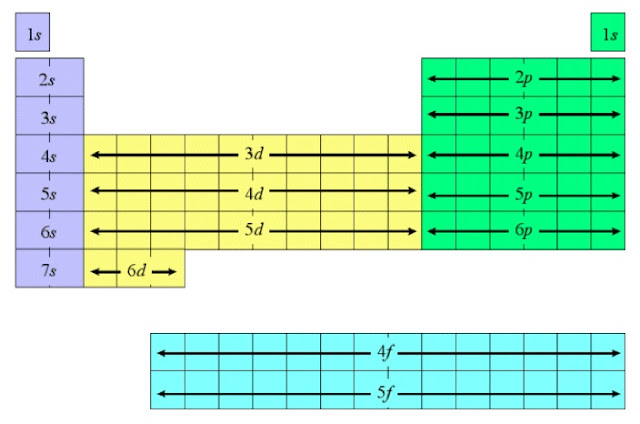 
Such as Lithium (Li) has an electron notation of 1s^2,2s^1. The same principal can be applied to any element on the table. The s defines the shape and movement of the electrons at this particular level, and finally the final one is the number of electrons occupying said shell. :max_bytes(150000):strip_icc()/PeriodicTableNaturalState-58b5d90c3df78cdcd8d0199b.jpg)
The first one means that the s class occupies the first energy level of the atoms electron cloud. The electron notation of hydrogen is 1s^1. Because of this it has only one orbital and one energy level for that orbital. Energy levels are specific orbitals that exist around an atom.įor instance, hydrogen, the first element on the periodic table, has only one valence electron. That means that the d class exists one energy level below the p class, and the f class exists two energy levels below the p class. The f and d classes are what one could call buried inside of the electron cloud (where the orbitals exist). Valence electrons consist of the s class and p class because these are the electrons furthest away from the nucleus and available for bonding with other atoms around them. An s class, p class, d class, and f class all of which have specific structures. Now there are four classes of these orbitals. But all physics aside, let’s just say that the electrons for our purposes swirl around the nucleus in what are called orbitals. In quantum mechanics, a branch of physics, the electrons are unstable particles whose existence varies from negligible to multiple places at once. Within an atom there is the dense nucleus which is positively charged and then negatively charged electrons orbiting around it. To understand valence electrons one must first understand the structure of an atom. 
These are known as quantum numbers, and they describe the identity of an element with the properties of its valence electrons. Not only do all of the elements hold a special place on the table, each element has a special number associated with it. It is arranged in order of atomic numbers and specific properties that identify each element into certain columns and rows known as groups and periods. The periodic table of elements consists of 118 of the purest substances known to man. So just use the orbital designation for quantum number shorthand.Periodic Table of Elements: Quantum Numbers Just using the n and ℓ we call this "orbital" a 1s orbital. The quantum numbers for this are n=1, ℓ=0, m ℓ=0, and m s=+1/2. So let's imagine a hydrogen atom in its lowest energy state. In this notation we simply state the principal quantum number n as a number.Ī letter is used to denote the ℓ term as letters s,p,d, and f. And really, just the first two are needed to identify the right KIND of orbital and energy level. When they are paired in an orbital you show both together as a pair, ⥮.Īs stated in the list above, we tend to just say "orbital" when referring to a specific set of quantum numbers. In electron energy diagrams we use and up arrow or better, a "harpoon" ↿ to depict a +½ spin state and a down harpoon ⇂ to depict a –½ spin state. For lack of a better term, "spin" was the winner for the name of this property. It was discovered much later because electrons were found to have another property that wasn't accounted for originally.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
